The latest patent statistics reveal valuable insights into Australia’s performance in medical technology and pharmaceuticals from 2020 to 2022.
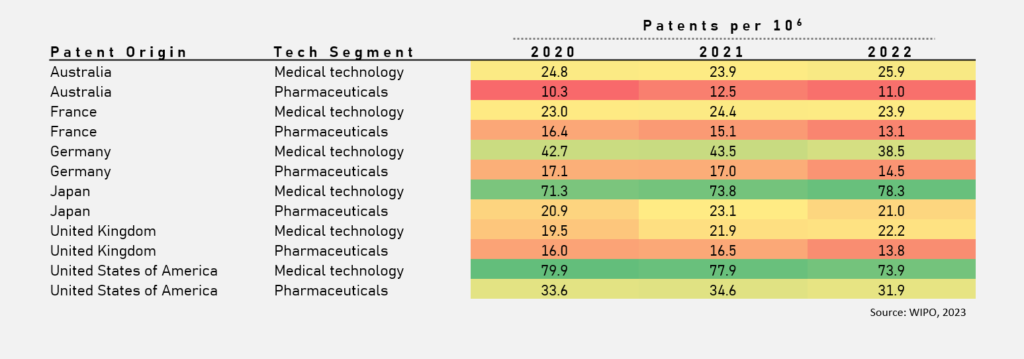
Australia’s medical technology patents rose from 24.8 per million in 2020 to 25.9 in 2022, demonstrating a steady commitment to innovation within this sector. This indicates a vibrant ecosystem where there’s active engagement in research and development, supported by strong scientific institutions and government initiatives.
Conversely, the pharmaceutical sector showed mixed results. This highlights the need for sustained investment and strategic support to maintain progress and encourage breakthrough discoveries.
Global Context
When compared to other countries, Australia’s performance is commendable, though it highlights areas for improvement, particularly in pharmaceuticals.
Japan leads in medical technology with 78.3 patents per million in 2022, demonstrating the success of its investment in high-impact health solutions. This indicates the importance of sustained government and private sector investment in fostering an environment conducive to innovation.
Germany and the United States also exhibit strong patent activity, showcasing the effectiveness of their nurturing of research commercialisation initiatives like a startup culture. In particular, the US had 79.9 patents per million in 2020 for medical technology, a benchmark for Australian innovators to aspire towards.
The Australian Advantage
Australia boasts a unique advantage in the medtech landscape, characterised by world-class research institutions and a young but growing startup ecosystem. Government initiatives, including the AU$15 billion National Reconstruction Fund, aim to bolster these industries, providing startups with crucial support for innovation and IP commercialisation.
Furthermore, Australia’s emphasis on collaboration among stakeholders has led to the establishment of various incubators and accelerators, facilitating the transformation of innovative biomedical ideas into market-ready products. This collaborative spirit is vital for driving growth and ensuring that Australian innovations can compete on the global stage.
Conclusion
For medtech founders navigating the dynamic landscape, these patent statistics serve as both a reflection of progress and a call to action. The upward trend in medical technology patents demonstrates that the Australian landscape is ripe for innovation. By prioritising strategic partnerships, committing to research, and focusing on effective IP commercialisation, founders can transform their ideas into solutions that significantly enhance health outcomes.
The Medtech Founder Playbook
Are you only starting your biomedical IP commercialisation journey? Consider taking the following key steps to ensure your patent strategy success:
1. Enhance Your Knowledge of IP
Understanding the intricacies of the patent process is essential. Investing time in educational programs can demystify the complexities of intellectual property (IP) protection. Workshops, online courses, and mentorship opportunities can empower you to identify patentable inventions early in your development process.
Medtech patents are highly valued by venture capitalists (VCs) because they indicate innovation and protects a startup’s unique technologies from competitors, reducing market risk. The patent application process can be daunting, but streamlining your approach can make it more manageable. Consider consulting with patent attorneys or IP experts who can guide you through the complexities and help you navigate potential hurdles.
2. Focus on Commercialisation Pathways
Understanding how to turn your patented technology into a viable product is essential. Seek resources and guidance on commercialisation strategies, including market analysis and business development. Success stories of other medtech innovations can inspire you and provide a roadmap for your own journey. Patents can facilitate strategic partnerships and licensing opportunities, and thereby helping to diversify revenue streams.
3. Engage with Regulatory and Reimbursement Frameworks
Familiarising yourself with the regulatory landscape and reimbursement pathways can significantly impact your innovation’s success. By aligning your product development with these frameworks, you can reduce uncertainty and increase the likelihood of successful market entry. This knowledge will also inform your patent strategy and enhance your overall business model.
At Pulse Economics, we can assist you in navigating your medtech commercialisation journey.

